Water, a limiting factor in lizard activities and distributionby Alice Loubet-Laouina
Published by the June 28, 2021 on 3:32 PM
Water is an increasingly scarce resource for us, so we can ask ourselves: When will it be for animals? How will they react: flee, adapt, evolve?
Predicting the effects of climate change on biodiversity is an ecological objective in conservation management. Here we are interested in water restriction role on activities and distribution on ectotherms with the mechanistic niche model.
Who am I

Photograph by Olivier Cardona, 2013My name is Tiliqua rugosa or Sleepy lizard.I am an ectotherm. I explain: I don't produce heat, so I need the sun to warm me up.
I live in Australia.
I measure 40 to 45 mm.
I live for 50 years in the wild!
I love to eat flowers, fruits, and the leaves of annual plants.
I can lose more than 70% of my body water despite being adapted to the desert.
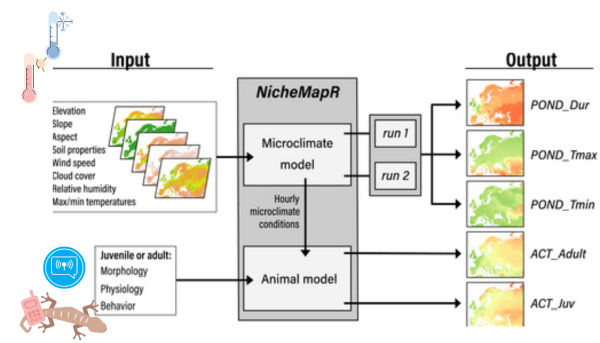
What is a mechanistic niche model?
So, the study scientists use this model with environmental and biological data of the Tiliqua rugosa. These data create Microclimate and Animal models. This allows us to understand how climate change affected species with a complex life cycle. This model makes it possible to make predictions on the distribution of animals. The same scientists used this method for another study (Enriquez-Urzelai et al. 2019).
Biological activities
When we talk about the biological cycle, we are talking about growth and reproduction activities. These activities require energy. Organisms have to make a trade-off between spending and storing energy.
The consequences of water restriction
When lizards only have access to water through their food or rainfall, their activities are reduced to the maximum to limit their dehydration and keep their energy. So Sleepy lizards hide in their burrows. They nest in a burrow up to 60 cm deep.
Water is a limiting factor in reproduction. According to two simulations, lizards reproduce little in the center and northeast of Australia.
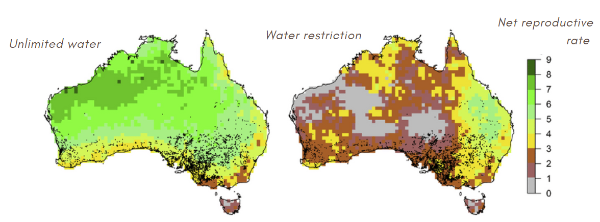
Estimation of net reproductive rateClimate change
To predict long-term evolution, we can use general circulation models in combination with mechanistic niche models. Therefore, it is interesting to compare different scenarios. Scenarios of the general circulation model1 can complement our niche model.
For the most part, water restriction is not a limiting factor for lizards. By running six scenarios under the assumption of water restriction, we find that the reproduction rate remains constant within the current ranges. The ACCESS 1.3 and GDFLCM3 Scenarios are exceptions.
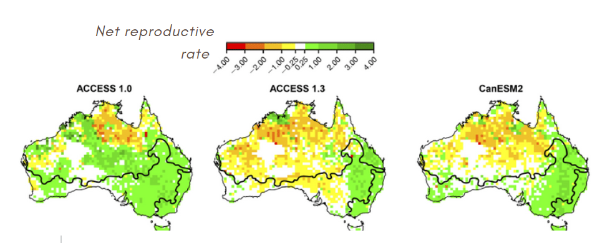
Estimation of different scenariosSpecies will be affected by climate change because it will depend on future precipitation patterns. Global warming could therefore cause increased dehydration regardless of rainfall variations.
1 General circulation model: statistics of several environmental factors allowing to make predictions according to different scenarios developed by countries.

This post is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.Spatial response of plaice and sole to climate changeby Arnaud Dupond and Alix Pascal
Published by the August 3, 2020 on 7:54 AM
Scientists admit that climate change is one of the main driving forces which change species distribution and abundance in many ecosystems.
In this case, a modification of abiotic variables can affect the “niche concept” and can also change species geographical distributions. For the marine environment, this notion of geographical dependence is important. Indeed, marine organisms show several distinct stages during their life and each of these stages evolve in a specific habitat.
The objective of this study was to use different models based on physiological aspects and environmental variables in order to estimate habitat occupation by plaice and sole under different climate change scenarios in the North Sea.
How to reach this objective?
To predict new habitats, researchers considered environmental variables and determined their effect on the food web, but also the effect of the food web on the water chemistry. To do that, the ecosystem model used functional groups of taxa. Their taxa are phytoplanktonic, planktonic and macrobenthic organisms. Some of them have a direct effect on the water chemistry and are regulated by other taxa. The food web is also used to quantify the availability of the habitat’s resources. Thanks, of this two types of model results fit more precisely reality of the distribution.
The sample strategy is built like this, data are collected daily on surface of 10x10km sin the North Sea.

Figure 1: Schematic simplification of the models used in the studiesResults of the study
The model of environmental variables shows the predictions for temperature and food conditions between 1989 and 2002. This data showed benthic production is concentrated along the southern coast during the year 1989, whereas in 2002 is concentrated in the Southern bight (figure 2).
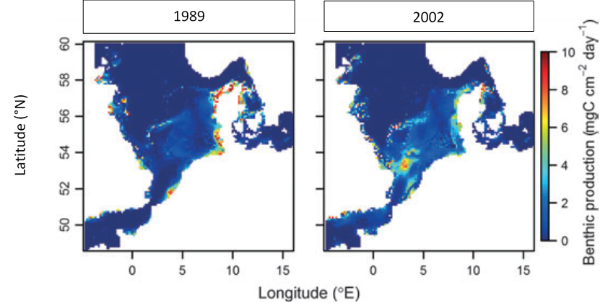
Figure 2: Comparison of the benthic production between 1989 and 2002 in the North SeaAn important fact is that the temperature rate inside of which the growth is positive will change with the size of the fish and according to abundance of food (the more food is abundant the higher rate of temperature is). Indeed, bigger fish need higher temperature to grow optimally. Figure 3 defines the areas of maximum potential daily growth of each class size of plaices in 1989 at the left, and in 2002 at the right.
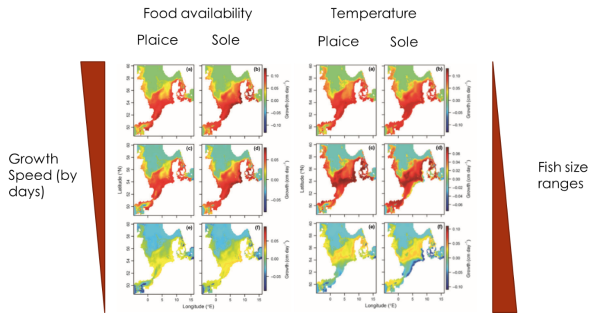
Figure 3: Comparison between the three size ranges of plaices and soles, of the speed growth in regard of two environmental parameters, the food availability and the temperatureThe results for maximum potential growth per day seem to give the same result as the estimate of average abundance. (Figure 4).

Figure 4: Comparison of the plaice and sole abundance distribution in the North SeaConclusion
For the plaice, migrations during different stages of life maximize their physiological performance during the summer season, in the winter, the adult’s distribution is determined by the best spawning habitat and shows maximisation of their fitness. Sole differs in their physiological traits and have a higher optimal growth temperature which explains the difference in life habitat. As for plaice, the area indicated high quality habitat for the different size class.
This study can predict the evolution of species distribution with a model of environmental changes and one of physiological changes but in our case, we can just explain data collected not the prediction made with the model.
Read the full study: Teal, L.R., van Hal, R., van Kooten, T., Ruardij, P. and Rijnsdorp, A.D. (2012), Bio‐energetics underpins the spatial response of North Sea plaice (Pleuronectes platessa L.) and sole (Solea solea L.) to climate change. Glob Change Biol, 18, 3291-3305. https://doi.org/10.1111/j.1365-2486.2012.02795.x

This post is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.A little bit of salt and heat... a good recipe for goby metabolism?by Maxime Deau, Quentin Garreau and Dorian Raoux
Published by the June 1, 2020 on 2:18 PM
As the literature shows, a variety of factors influence the well-being of fish populations. For example, we know that some fish may or may not be very sensitive to changes in the conditions of their living environment (water temperature or salinity). These changes can affect their metabolism (reduced fertility, growth, etc.) or even, in the worst case, lead to the death of individuals. The goby (Pomatoschistusmicrops) (Figure 1), a relatively tolerant species and an essential central link in the food web is one of the species studied in the observation of the impact of these changes on the fish fauna in the Minho estuary in Portugal (Figure 2).
In this study, the researchers were able to model the evolutionary dynamics of p.microps populations based on models that take into account different parameters of goby's life cycle like fertility, mortality, migration rate and the effect of environmental parameters such as salinity and temperature. The aim of these models is to describe the evolution of the different life stages of this fish by establishing the possible impacts of climate change on their metabolism. In this framework, they studied both the impacts of temperature and salinity and combined the impact of both.
It has been noted that salinity directly influences the metabolism of individuals. Indeed, it plays a particular role in the survival of many aquatic organisms but also on their growth (strong allocation of energy to osmoregulation; Rigal, F. and al.,2008). Therefore, it plays a role in the growth of the goby as well as indirectly on its prey. The latter will be less available, which implies a higher energy expenditure for predation. However, this species resists large variations in salinity (0 to 51 psu). For temperatures, the impact is more diverse. Since the goby does not thermoregulate, its metabolism is directly influenced by the temperature of the environment. In addition to its significant effect on pregnancy, it also has an impact on migration, reproduction, recruitment and mortality. (Sogard, 1997; Hurst et al., 2000; Hales and Able,2001; Hurst, 2007; Jones and Miller, 1966; Claridge et al.,1985; Wiederholm, 1987).
Regarding the goby’s responses to these parameters, the research team has implemented them in the model, running different scenarios (salinity and temperature variations). Temperature and salinity variations studied separately led to population crash, except for a salinity lower than the current state ( -5psu). However, the combination of the two variables gave scenarios showing an increase in the population when the salinity was -5 psu, with temperatures ranging from +1 to +3°C, with an optimum at +2°C (see figure 5).
For example, in extreme temperatures, the fish activity will be greatly reduced, which will imply a decrease in the search for preys or sexual partners, causing feeding and mating problems (predation of eggs by males (Magnhagen, 1992). However, a slight increase in temperature could cause a longer reproduction period, allowing for a greater number of offspring to be generated. It has also been noted that with an increase in temperature, there is a delay in the breeding period, leading to the appearance of offspring in a period that may be less favorable for their proper development (early winter/lower metabolism).In conclusion, climate change, through its effects on water temperature and salinity, will have a significant impact on common goby populations. Indeed, these parameters have a great influence on the metabolism of these fish (whatever their stage of development).In many scenarios, increases in temperature and salinity can cause crash populations. But beware, in some cases (increase in temperature and decrease in salinity) the population of Pomatoschistus microps would tend to increase. Even if this scenario seems favorable for this species, some others will suffer. in fact, a study conducted on Arctic fish species has confirmed these trends
It is therefore clear that climate change affects population dynamics by changing fish environment and impacting their metabolism. It’s therefore important to continue this kind of study to have a better idea of these repercussion on a global scale. We are largely responsible for climate change, so it is up to us to make sure that we limit our impacts. Here is a link that will teach you how to reduce your carbon footprint through 20 examples of simple everyday actions: http://www.globalstewards.org/reduce-carbon-footprint.htm
Other cited studies:
Claridge, P.N., Hardisty,M.W., Potter, I.C., Williams, C.V., 1985. Abundance, life history and ligulosis in the Gobies (Teleostei) of the inner Severn Estuary. J.Mar. Biol. Assoc. U. K. 65, 951–968.
Hales, L.S., Able, K.W., 2001.Winter mortality, growth, and behavior of young-of-the-year of four coastal fishes in New Jersey (USA) waters. Mar. Biol. 139, 45–54.
Hurst, T., 2007. Causes and consequences of winter mortality in fishes. J. Fish Biol. 71, 315–345.
Hurst, T.P., Schultz, E.T., Conover, D.O., 2000. Seasonal energy dynamics of young of the year Hudson River striped bass. Trans. Am. Fish. Soc. Taylor & Francis 129, 145–157.
Jones, D., Miller, P.J., 1966. Seasonal migrations of the common Goby, Pomatoschistus microps (Kroyer), in Morecambe Bay and elsewhere. Hydrobiologia 27, 515–528.
Magnhagen, C., 1992. Alternative reproductive behaviour in the common goby, Pomatoschistus microps: an ontogenetic gradient? Anim. Behav. 44, 182–184.
Rigal, F., Chevalier, T., Lorin-Nebel, C., Charmantier, G., Tomasini, J.-A., Aujoulat, F., Berrebi, P., 2008. Osmoregulation as a potential factor for the differential distribution of two cryptic gobiid species, Pomatoschistus microps and P. marmoratus in French Mediterranean lagoons. Sci. Mar. 72, 469–476.
Sogard, S.M., 1997. Size-selective mortality in the juvenile stage of teleost fishes: a review. Bull. Mar. Sci. 60, 1129–1157.
Wiederholm, A.-M., 1987. Distribution of Pomatoschistus minutus and P. microps (Gobiidae, Pisces) in the Bothnian Sea: importance of salinity and temperature.Memoranda Societatis pro fauna et flora Fennica 63, 56–62.
This post is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.Corals and algae, a relationship in danger: a model to predict their future! by Clara Dignan, Anna Gago and Anabelle Leblond
Published by the March 2, 2020 on 2:14 PM

Bleached branching coral (foreground) and normal branching coral (background). Keppel Islands, Great Barrier ReefCorals that come together to form coral reefs are shelter to 25% of our planet's marine life according to the WWF. This biodiversity is fundamental. It’s both a source of income and food, and it provides irreplaceable services to humanity. But today coral reefs are in danger. They are directly threatened by global warming. In forty years, 40% of the reefs have already disappeared and scientists agree that if nothing is done by 2050, all of them will be gone (Coral guardian).
Coral polyps and algae, an endosymbiotic relationship
Coral bleaching has now become a major global concern for the future of coral reefs. Temperature rise appears to be one of the main causes of bleaching, affecting growth, feeding and other ecological processes on reefs. This bleaching phenomenon is due to the expulsion of zooxanthellae, the symbiotic microalgae living in the tissues of the polyps (the coral is made up of a colony of polyps that participate in the making of its skeleton). These unicellular algae carry out photosynthesis and provide, for the most part, the energy that corals need to develop and grow. Exchanges between the polyp and the zooxanthellae mainly concern nitrogen, phosphorus, carbon and biosynthetic intermediates. The presence of zooxanthellae being responsible for the color of the colonies, bleaching is therefore the symptom of a coral which is no longer in symbiosis, which generally results in the death of the coral.
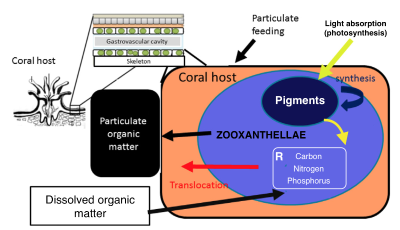
The coral-symbiont relationship and its interaction with the overlying water column.Prediction models
Since few year, scientists analyze corals and try to predict their bleaching evolution. In this aim, a collaboration between several organizations such as CSIRO have set up a first hydrodynamic, sedimentary and biogeochemical model called: « eReef ». This model simulates the environmental conditions as the temperature, the background light and the organic nutrient concentration of the Great Barrier Reef at several scales. It allows accurate prediction of factors influencing coral processes from satellite remote sensing images.
However, for more representative modelling, it is necessary to apply models that take into account the coral-symbiont relationship and the stress related to environmental variations. In this framework, Baird et al. have developed a model which, in parallel to the environmental conditions obtained from the « eReef » model, also takes into account essential parameters in the symbiotic process such as biomass and growth rate of zooxanthellae, pigment concentration, nutritional status as well as tolerance characteristics such as sensitivity to reactive oxygen concentration (oxidative stress).
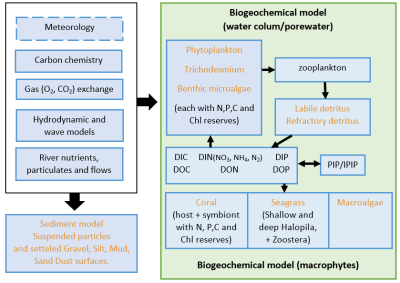
The eReefs coupled hydrodynamic, sediment, optical, biogeochemical model. Orange labels represent components that either scatter or absorb light levels. (For a better understanding of the colour used and the abbreviations, the reader is referred to the web version of the article)Take home message
This coral bleaching model applied under realistic environmental conditions has the potential to generate more detailed predictions than satellite coral bleaching measurements. In addition to predicting coral bleaching, this model will now make it possible to evaluate management strategies, such as the introduction of temperature-tolerant individuals or species or localized shading.
Nevertheless, this model is still too simplistic to make real predictions. It is based only on the process of a single type of coral and macro-algae and does not take into account all phenomena related to bleaching. It is therefore seen as a step forward for science that could allow for future reevaluations of the effects of bleaching.
Bibliography
What does the future has in store for red salmon in a context of global climate change?by Camille Sestac and Amandine Tauzin
Published by the November 1, 2019 on 1:18 PM
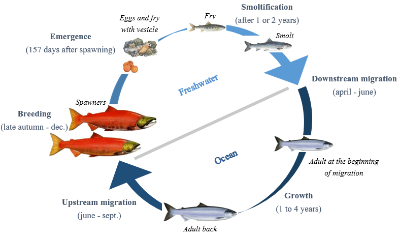
Pacific salmon have extremely complex life histories and may be threatened by global climate change, as Peter S. Rand and colleagues investigate in their recent study.
Among all species, fishes must adapt to face disruptions caused by global climate change. Sockeye salmon (Oncorhyncus nerka), an anadromous species of salmon found in the Northern Pacific Ocean and rivers discharging into it, has a complex life cycle. As a migratory species, their energetic demands are high during spawning migration. Climate change might have important impacts on populations and their migration via variation of river discharge, increase of water temperature and decline of growth conditions. Aiming to better understand the impacts of these disruptions on the migratory performance of this species of salmon, Peter S. Rand from Wild Salmon Center teamed up with researchers from British Columbia. Their goal is to evaluate the effects of past and future trends in river discharge and temperature on the migratory performance of Sockeye Salmon in the Fraser River.
In a context of global climate change, it is crucial to understand the effects of disruptions on ecosystems and the populations living in them. Indeed, it is important to know the impacts of these disruptions on every stage of their life cycle (the juvenile freshwater period, the estuarine period, and the subadult marine period) so that we can maintain the populations stock. It’s especially important for fishery management because the fishing quota has greatly increased over the last decades and has threatened populations of Pacific salmon, particularly during their spawning migration. That’s why with three main objectives, these scientists used analysis to improve the understanding of how changes in river conditions can affect the energy use and the mortality rate in Sockeye salmon population. To do so, they used several models: one to search a link between energetic conditions of individuals and en route mortality, one to simulate the energy use during spawning migration and one to hindcast and forecast energy use by simulating fish’s behaviour and migration conditions (for more information, a tip, read the article!).
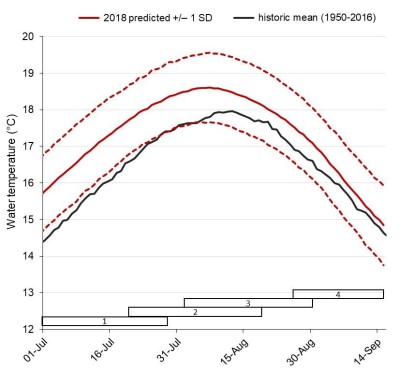
Long-range forecasts of lower Fraser river temperature during the summer of 2018Using these friendly models, Rand and his colleagues proved that energy reserves and energy depletion of early Stuart Sockeye salmon are major factors that can affect their ability to reach their spawning grounds. They also stated that energy depletion is a function of both river temperature and discharge. Therefore, this population is structured by condition-dependant mortality. Nevertheless, this group of researchers brought to light a mechanism that allows fishes to cope with some environmental variability, providing a certain degree of resilience over time. Therefore, even if energetic demands and migration mortality increase as a result of exposure to warmer temperatures, it will be compensated by reduced time travel to the spawning ground as the river flow will be lower.
However, increase of temperature means increase of diseases appearing and developing and that stress added may be a direct cause of increased mortality during migration. Finally, as if it wasn’t already bad enough for our salmons, ocean productivity can be affected by climate change and thus affect their river migration success. In fact, this can lead to a decrease of body size and body energy content. It implies that individuals will start their migration with lower energy densities and will be more likely to exhaust their energy stock before even reaching the spawning grounds.

Salmon jumping over a weirAccording to the US-Canada Commission, a 21° C temperature spike was measured on the Fraser River in 2009. However, sockeye salmon show signs of physiological stress and migratory difficulties above 19°C and from 20°C, the first signs of illness and death appear. But migration of Sockeye salmon is not only threatened by climate change. In fact, migration of salmon specially is impacted by humans or natural obstacles. Dams and weirs form large obstacles for this migratory species and can be very difficult to cross. Many studies have already proved that this kind of obstacles, even when equipped with crossing devices, delay their migration and thus jeopardize their reproduction. This can lead to a decline of the population and in some cases to its extinction, as it happened in Belgium.So, whilst some questions have been answered, it seems that more studies need to be carried out to improve our knowledge about the impact of global change which seems to be another sword of Damocles hanging over the head of Sockeye salmon.
Cited paper: Rand, P.S. et al. (2011) Effects of River Discharge, Temperature, and Future Climates on Energetics and Mortality of Adult Migrating Fraser River Sockeye Salmon. Trans. Am. Fish. Soc. 135(3), 655-667. https://doi.org/10.1577/T05-023.1
Featured images: Life cycle of Sockeye salmon by Camille Sestac, graph from https://www.pac.dfo-mpo.gc.ca/science/habitat/frw-rfo/index-eng.html , Sockeye Salmon from www.ryanvolberg.com

This post is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.
Organisms and their environment: Dangerous liaisonsby Marius Dhamelincourt and Charlie Sarran
Published by the October 14, 2019 on 9:15 AM
Preface
Whatever the incredulous think, organisms are necessarily linked to their environment to survive, science says. However, this relation, unstable, can be problematic for those concerned when variations overtake their acceptable thresholds. Although often natural, these variations can be exacerbated by anthropogenic actions, like putting fish on a grill.
Global changes are often reduced to temperature increases, illustrated in the media with alarming news about ice melting and forest fires. While many people thus omit the complex mechanisms behind this black box, the necessity of a more “polar bear’s” respectful way of life is commonly accepted. In order to better understand how to respect such adorable creatures, scientists need to investigate their relationships with the environment.
Chapter 1: Shells under investigation
In order to scrutinize these relationships, selected species must be accessible, easy to catch/manipulate and in sufficient number. For instance, the study of the great white shark aggressivity over humans would require too many intern’s sacrifices. In response to this challenge, a valiant research team from Germany looked for the importance of these relations by studying in the Rhine a remarkable (body and soul), accessible, cheap and lovely species: Corbicula fluminea, a shell. This study is related to the mass mortalities events of this species, which occurred in the summer, especially in that of 2003. Their investigations aim to understand how these organisms are linked with their environment, and their reactions to changes.
Chapter 2: Shells cooking in science
Corbicula fluminea shellsMany tools exist to perform this type of search. Field searches can involve the scientist’s life (be bitten by a pigeon is a terrible experience…) and obstruct a long-term individual experiment. Now that researchers have selected the perfect organism, they must choose an appropriate way to analyse their problem. For this shell, scientists chose to use a modelling approach, a method dark and full of terrors. More precisely, they modelled several aspects of the metabolism of this organism at different scales: individual and population levels, using respectively DEBM (Dynamic Energy Budget Model) and PSPM (Physiologically Structured Population Model). This method, widespread in ecology, consists to “simulate the annual growth in length and mass and the reproductive success under different environmental scenarios”. This approach is suitable because an organism can respond differently relatively to their peers. Such fact can be proved by looking at many places and species, humans included… Ultimately, scientists aim to better understand the complex relation between the energy available in the environment and its utilization by shells.
Chapter 3: Corbicula’s deadly summers

Heat waves are often responsible for changes that every scientist can observe on living organisms. For instance, it is known that coral reefs are affected by increased temperatures, as shown in an article published in “Free Radical Biology and Medicine”. Many other examples such as lobster’s behavioural response to boiling water could be developed. Regarding our shellfish, scientists found an interesting pattern comforting our previous remark: temperature causes shell’s mortalities… Oh wait, no, it’s more complicated.
In fact, mass mortalities events were probably related to a melting pot of many events like temperature increases and/or starvation. Moreover, these situations are also in relation with individual conditions. Indeed, researchers hypothesized that a combination of factors (biotic and/or abiotic), usually non-lethal under regular summers, can be problematic at high temperatures. Unfortunately, models developed were not able to explain completely the observed mass lethal events.
Chapter 4: Life is not so simple
Researchers finally enhance the comprehension of population dynamics, enlightening its complex mechanisms. However, in such cases, wishing to be exhaustive is useless and unproductive, like politics. That is why scientists look for compromise between easy-to-use and complicated (highly realistic) tools. For example, the authors of the Corbicula’s study proposed that it could be interesting to test other parameters, such as parasitism.
To put it in a nutshell (you got it, right?), things are not always what they seem to be, even in environmental studies. Main hypotheses are not always validated, and measures considered can be only a part of a more complex system, or sometimes even unappropriated. On the flip side, model’s development can help to understand the life cycle of organisms like Corbicula, thus helping to manage populations concerned.

This post is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.Global change and climate-driven invasion of the Pacific oyster along European coasts: a bioenergetics modelling approachby Mélanie Gouaux and Lise Guégniard
Published by the January 8, 2019 on 3:20 PM
Environmental changes such as seawater warming, and coastal eutrophication have an impact on breeding, larval survival and recruitment of marine benthic species. Global change induces changes in the natural distributions of native species and facilitates the spread of non-native species. Nowadays, the spread of non-native species in marine ecosystems around the world is one of the most serious environmental concerns. In receiving ecosystems, consequences of biological invasions are readily identifiable when invasive species are ecosystem engineers such as polychaetes or bivalves.
Scientists of the French Research Institute for the Exploitation of the Sea and of the university of Nantes investigated how global change relates to the invasion of European coasts by a non-native marine invertebrate, the Pacific oyster Crassostrea gigas.
This species was introduced on the European coasts of the Atlantic at the end of the 19th century for shellfish culture purposes and is the main oyster species farmed in Europe today. In recent decades, the Pacific oyster has acquired invasive species status with the expansion of its biogeographic distribution along the northwestern European coast beyond its initial zone of introduction into sites breeding. Bourgneuf Bay on the French Atlantic coast was considered as the northern boundary of C. gigas expansion at the time of its introduction to Europe in the 1970s. From this latitudinal reference, variations in the spatial distribution of the C. gigas reproductive niche were analysed along the northwestern European coast from Gibraltar to Norway.

A bed of Pacific oyster Crassostrea gigas in the Netherlands - Bas Kers - CC BY-NC-SA 2.0Mechanistic models are valuable tools for this purpose, and modelling approaches are useful for gaining a quantitative understanding of the effects of environmental changes on marine communities, and predicting their responses to projected climatic trends.
The use of the IBM and DEB models has shown results at different scales, at the individual scale, at the Bourgneuf Bay scale and at the European scale.
At the individual level, the results showed interannual variability of the dry flesh mass (DFM). A loss of DFM is explained by a spawning event. There has been a significant increase in DFM and the number of oocytes in recent years. To explain these results, they studied the relationship between the environmental conditions in late spring and the characteristics of the oyster. Then they achieved positive relationships between DFM and phytoplankton. Likewise, between the number of accumulated oocytes and phytoplankton. Here, phytoplankton seems to be the cause of this increase of DFM and the number of oocytes in recent years for this species of oyster. Here, phytoplankton seems to be the cause of this increase of DFM and the number of oocytes in recent years for this species of oyster. Moreover, according to the individual model, the higher the temperature of the sea surface, the earlier the clutches (June-July). While a cooler sea surface temperature will result in late laying (August-September).
Then, they applied the same models to the bay of Bourgneuf. The results showed an increase in the temperature of the sea surface in the bay, but also the effects of the temperature of the water on the laying. Indeed, as at the individual scale, the high-water temperatures lead to prose spits and vice versa.
Results at the European level showed a change in the geographic limit of spawning habitat, regardless of phytoplankton concentration. In 1986, the limit was located at the level of the Loire estuary with a south-north spawning gradient, earlier in the south of Europe. In 2003, this limit moved completely to the north of Europe. This change can be explained by the global warming of the waters.
Other studies have highlighted other results. Indeed, using a Degree / day model, Ifermer has demonstrated the importance of the nutrient pool for egg laying. Indeed, in recent years, laying is actually late because of the low nutritional value of phytoplankton. The warming of the waters would therefore cause the loss of the oyster's nutrient pool and thus a delay in laying eggs.

This post is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.Temperature-dependent body size effects determine population responses to climate warmingby Alison Arraud and Laura Duran
Published by the July 5, 2018 on 1:55 PM
Up to now, neither the size nor the stage of the individual were considered to studying the population responses to climate warming. On 2014, a scientific group proposed another way to understand the temperature effects on fish populations. They improved the interaction effects of temperature-dependence with the size and the stage of fish on their energetic thresholds responses. Energetic thresholds themselves act on the dynamic of stade-structured population (e.g. parr, smolt, adult).

Flathead mullet (Mugil cephalus) - Roberto Pillon - CC BY 3.0 UnportedFinally, this study found that increasing temperature could redistribute biomass across life stages and modify the regulation of the population by reworking the intra-specific competition. Other studies have shown that high temperature during ontogenesis can accelerate the development and growth of individuals or, give individuals of smaller sizes early maturation.
This study points out the importance of taking into account the interactions between temperature and size-specific (maturing, reproduction, etc.) that will lead to a set of behavioral responses that have consequences on the structuring of a population. This is all the more important in the context of global warming.

This post is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.